[Trade Journal]
Publication: Electrical World
New York, NY, United States
vol. 5, no. 22, p. 216, col. 1-2
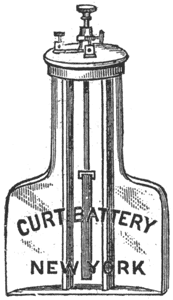
The "Curt" Battery.
The Grenet battery, which has been so long and favorably known, while giving good results in its way, is nevertheless, as ordinarily made, open to some objections, and it was to remove these that the battery shown in the annexed engravings has been designed.
In the first place it will be noticed that though occupying no more space in the box than the Grenet round cell, the "Curt."' No. 1 contains nearly twice as much fluid as the other, and exposes a surface of electrodes, both carbon and zinc, which is equal to that of the No. 2 Grenet; hence there results a current of greater quantity and duration from a battery of equal outside dimensions.
Added to this the lower halt of each carbon is corrugated, and to prevent corrosion of the broad metallic flanges to which the carbons are attached, the upper ends are saturated with paraffine. The brass flanges, where they make connection with the carbons, are covered with a sheet of platinum so that no corrosion can take place, leaving the contact unimpaired. The other surrounding parts are given a coat of asphalt varnish. The carbons are not soldered, but screwed on, and a new one can be attached at a moment's notice.
Another improvement introduced is the hard rubber cap, which does away with all metallic mountings around the neck of the jar. In order to accomplish this, the "Curt" battery is provided with a groove around the upper part of the jar, into which fits a flexible rubber ring. This holds a solid, hard rubber cap firmly, and closes the jar hermetically.
 |
As the metallic mounting is done away with, any fluid destructive to metals may be used, such as bisulphate of mercury solution, producing a very continuous current for electric light or cauterizing purposes. In the ordinary Grenet battery this fluid cannot be used without destruction to the metal mounting on top of the jar.
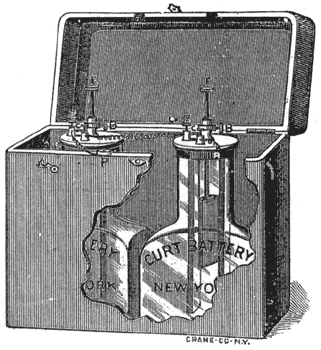 |
Another point worthy of note is the rod supporting the zinc. This, for convenience of transportation, is made in two sections, the upper one screwing into the lower.
Two of these cells, which are made by Mr. Curt W. Meyer, of this city, are calculated, to furnish the exact amount of current to light a 3-ohm incandescent lamp for medical purposes, and develop its full illuminating power, without danger of destroying the carbon filament of the lamp from too strong a current.
