[Trade Journal]
Publication: Electrical World
New York, NY, United States
vol. 11, no. 24, p. 307, col. 3
The Samson Primary Battery.
We illustrate on this page the Samson battery for open circuit work, recently brought out by the Electric Gas Lighting Company of Boston, which has the exclusive control in America, and the tests of which show some remarkable results. One leading feature of the battery is the carbon, made up in the form of a corrugated vase, giving an unusually large surface. This is almost entirely inclosed by the zinc in the form of a thin sheet, to which a terminal like the ordinary zinc rod is attached. Contact between carbon and zinc is prevented by little earthenware buttons set in the carbon, and the whole is inclosed in a glass jar of the familiar shape. A solution of sal ammoniac is used as the electrolyte, and as soon asset up the battery gives out its current for work. The top of the battery is protected by a rubber cover fitting snugly around the teeth or flutings of the carbon cup. This battery has been in use, we are informed, for two years, on the French Government telegraphs, with excellent results. We give below a report from Prof. A. E. Dolbear, of Tufts College, on the battery:
TUFTS COLLEGE, May 18, 1888.
Report on the performance of four cells, examined for their E. M. F., Internal Resistance and Current Strength.
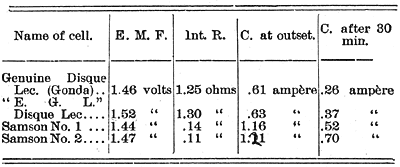 |
The cells were all put in circuit with a resistance of 1.1 ohms, and kept on that circuit for the above measure of current. The very small internal resistance of the two "Samsons" enables them to give a current very nearly twice as great at the beginning as either of the others, while the "Samson" No. 2 gave a stronger current (.70 ampère) after half an hour's short circuit than either of the Disque Leclanché cells at the beginning. It should be remarked here that the Samson No. 1 was a smaller cell than the No. 2, and, of course, would not be expected to give the current of the large one. Chloride of ammonium batteries have this advantage over most other cells, that after working and a partial exhaustion they will recover their original power if left unused for a time. The length of time needed to fully recover depends upon the draught made upon them, but when the period of use is short, as in bell ringing, gas lighting, etc., there is always ample time for full recovery. It appears, therefore, that the Samson cells are much more energetic than the ones they were compared with; so much so, that one of them is about as good as two of the latter kind for such service as the production of strong currents or strong magnetic effects. The device of making the carbon cylindrical and fluted enables them to contain a large quantity of the binoxide of manganese, and presents a large surface to the solution. The shape of the zinc and its proximity to the carbon is another advantage for such work. I have also tested the cells for telephone work, and find them very superior. Their small internal resistance enables them to give a stronger current through the induction coil than any Leclanché cell I have ever tested before. This amounts to 25 or 30 per cent. The cell meets my commendation.
 |
| The Samson Battery. |
